 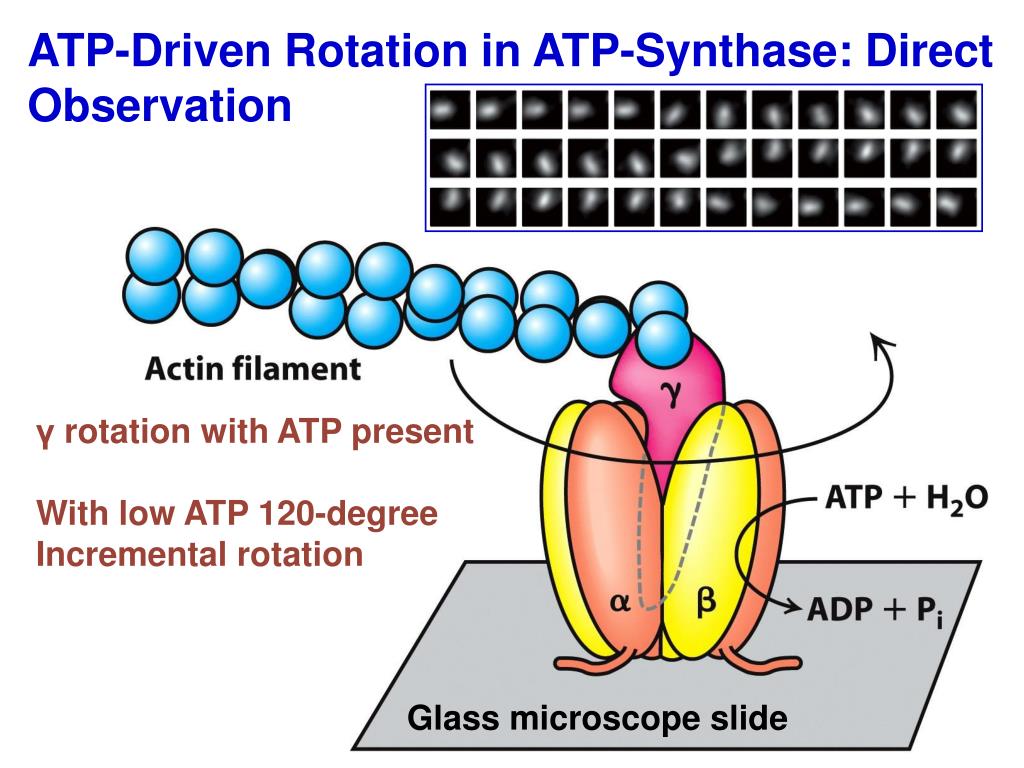
The diagrams below helps illustrate the proton gradient I am referring to. The ion's charge is used to drive their individual processes, like making ATP, and this cycle fulfills both the protein's and the ion's wants. Proteins in the membrane utilize the ion's need for help and tendency to want to go across the membrane by making it useful for them. Since H+ protons are ions pumped across, they need a little help getting back to the other side of the membrane. With an abundance of protons on one side of the membrane it will have a strong positive charge and a bunch of protons ready to leave and go back across the membrane - nature will always try to reach equilibrium. In the Electron Transport Chain, protons are pumped to one side of the membrane creating a transmembrane potential. Note that 'consuming' H in the matrix increases the 'proton' gradient across the IMM, so this helps with energy production. As far as I can tell the hydrogen ions come from hydronium (HO), which is naturally present in all aqueous solutions. In the case of ATP, while there are other forces that drive the synthesis of ATP we will focus on the proton gradient. This 'proton' gradient is what drives oxidative phosphorylation. The charge gradient is formed and this is how the energy in cells can be stored. A cell membrane is made up of a semi-permeable substance (lipids) that prevent the free-flow of ions across it because ions can not freely move from one side to the other, it creates charge on one side and a lower charge on the other. The reason we can do this is that the basic structure of the cell membrane. To help visualize cell potential in a membrane, we can actually view the cell membrane as a parallel plate capacitor with a dielectric. The downhill flow of protons from an area of high concentration in the inner membrane space to an area of low concentration in the. Protons flow downhill from the inner membrane space through the ATP synthase protein. In this section, I will be talking about how electric potential in a membrane works and then more specifically on how the electric potential within the mitochondrion drives the synthesis of ATP. The ATP synthase enzyme uses the proton gradient formed by the electron transport system to drive the synthesis of ATP. The flow of protons is motivated by the flow of electrons along a series of stations leading up. Usually when we think electric potential a battery will pop into our mind, not a cell membrane. It is driven by a flow of protons that pass through its shaft. The H+ ions provides the protein with the energy to twist and squish together ADP and a Phosphate group to. The electron transport chain forms a proton gradient across the inner mitochondrial membrane, which drives the synthesis of ATP. Mitchell (1961) postulated that Nature uses protonic batteries to drive ATP synthesis and. Here, light energy drives electron transport through a proton pump and the resulting proton gradient causes subsequent synthesis of ATP. Electric potential across membranes is an incredibly interesting concept to consider. ATP synthase will twist as the proton moves through. Introduction ATP is the common energy currency of the cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
